- #FLORIDA CLINICAL LABORATORY PERSONNEL LICENSE VERIFICATION VERIFICATION#
- #FLORIDA CLINICAL LABORATORY PERSONNEL LICENSE VERIFICATION CODE#
- #FLORIDA CLINICAL LABORATORY PERSONNEL LICENSE VERIFICATION LICENSE#
Florida’s Agency for Health Care Administration (AHCA) expects to roll-out notifications regarding the change in state licensure requirements to currently licensed clinical laboratories in approximately two weeks and will post notice on its website. § 483.091).īeginning July 1, 2018, clinical laboratories and stakeholders will be able to provide their laboratory services in Florida or to Florida healthcare providers as long as they meet federal CLIA certification requirements. Our state licensing map was built to help medical lab scientists find all of the information they need to attain the necessary state personnel and facility.
#FLORIDA CLINICAL LABORATORY PERSONNEL LICENSE VERIFICATION CODE#
Code 59A-7.024) or (2) receives a specimen for examination from a clinical laboratory located within the state of Florida (Fla. Furthermore, an out-of-state laboratory testing specimens derived from the state of Florida is also required to obtain Florida state licensure if: (1) the out-of-state laboratory maintains an office, specimen collection station or other facility within the state of Florida (Fla. government (2) a clinical laboratory that only performed waived tests or (3) a clinical laboratory that was operated and maintained exclusively for research and teaching purposes that did not provide services to patients.
#FLORIDA CLINICAL LABORATORY PERSONNEL LICENSE VERIFICATION LICENSE#
Section 97 of SB 622, approved by the Governor on March 19, 2018, repeals the entirety of Chapter 483, Part I of the Florida statutes, and therefore, in tow, eliminates section 59A-7.024(1) and as well as all other corresponding regulations.Ĭurrently, all clinical laboratories providing services within the state of Florida must maintain a state license unless you were either: (1) a clinical laboratory operated by the U.S.
Effective July 1, 2018, Florida’s recent legislation ( SB 622) repeals the entirety of Chapter 483, Part I of the Florida statutes, and in doing so removes the state licensure requirement for clinical laboratories operating in-state and out-of-state. Written notification includes email, fax or hard copy letter.The Florida State Legislature has decided to eliminate its state licensure requirement for clinical laboratories. If the CMS-116 is used in lieu of written notification, it must be completed in it entirety to be accepted. SUPERVISED CLINICAL EXPERIENCE I have read and understand Rule 64B4-2, Florida Administrative Code (F.A.C.), which states, in part: An intern shall be credited for the time of supervision required by s.
#FLORIDA CLINICAL LABORATORY PERSONNEL LICENSE VERIFICATION VERIFICATION#
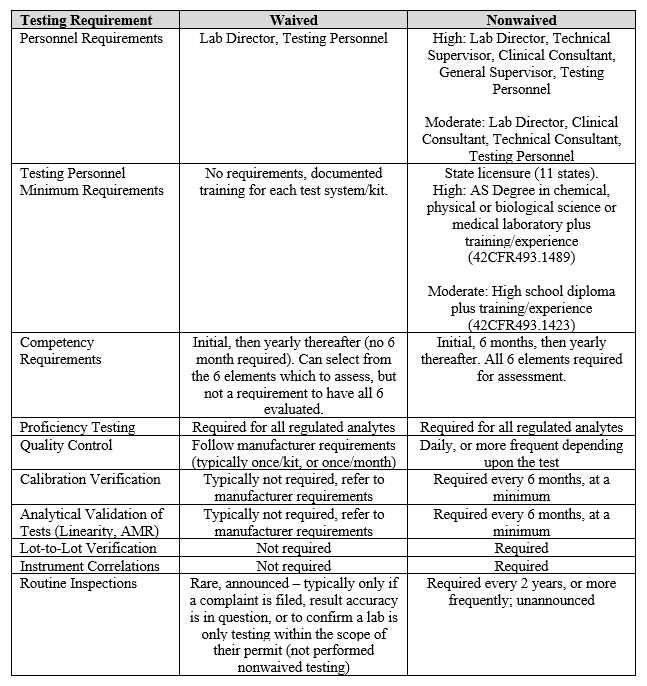
NOTE: A Form CMS-116 can be submitted when written notification is required. The Florida Board of Orthotists & Prosthetists was established to ensure that the citizens of Florida receive appropriately designed, fabricated and fitted orthotic and prosthetic devices from licensed providers. License Type State License Number Supervisors licensed outside of Florida must provide a license verification 2. Reinstatement if there is no gap in service (please call our office if you are unsure if there is a gap).Change in address, whether street address or mailing address.Written notification of changes (always provide the CLIA ID number):.Reinstatement Application if there is a gap in service.Submit a new Federal CLIA application (FORM CMS-116) for:.There are no requirements for directors of waived laboratories.Waived laboratories are not inspected except on a random basis for the CLIA program.Follow manufacturer's instructions and document tests performed.CLIA Certificate of Waiver renewed every 2 years.Requirements for Laboratories Performing Only Waived Tests: Conduct testing that is considered non-technical requiring little or no difficulty.Pose no reasonable risk of harm to the patient if the test is performed incorrectly.Employs methodologies that are so simple and accurate as to render the likelihood of erroneous results negligible.All facilities performing any clinical Laboratory testing are required to obtain a CLIA certificate. The Centers for Medicare and Medicaid Services (CMS) administers the CLIA Laboratory certification program in conjunction with the Food and Drug Administration (FDA) and the Centers for Disease Control and Prevention (CDC). Blood glucose- devices cleared by the Food and Drug Administration of home useįederal Requirements for a Certificate of Waiver:.


